EXTERNAL LINK
You are about to leave Pajunk.com. This link is provided strictly for information sharing purposes.
Pajunk GmbH assumes no responsibility for the quality, content, nature, or reliability of any linked site.
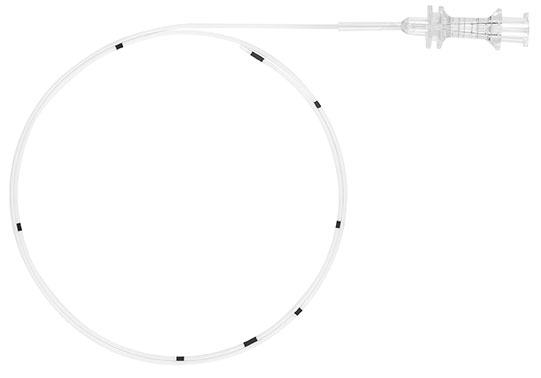
The InfiltraLong catheter developed by Pajunk is a self-contained system for continuous infiltration analgesia at the surgical site. The design of the catheter allows a smooth and uniform targeted administration of local anaesthetic. This allows to optimise patient analgesia and improve outcomes while reducing opoid consumption.1
The InfiltraLong catheter is the only wound infiltration catheter that can be combined with all commercial pumps. The wide range of catheter and perforation lengths available makes it suitable for many different requirements and areas of use.
In comparison with three other commercial catheters the InfiltraLong performed extremely well because of its numerous fine infusion perforations, which are closely and evenly spaced in the perforation segment.2 This ensures an even distribution of the anaesthetic and a earlier mobilisation3. The
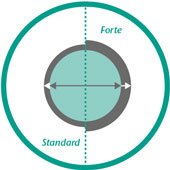
InfiltraLong catheter is available in a standard version as well as the Forte version with an extended wall thickness.
The InfiltraLong sets come in many different configurations, including basic accessories like introducer needles and filter, but also full sets with a pump.





| Item description | Item no. LUER | Purchase Unit |
|---|---|---|
|
InfiltraLong 420 / 19G x 420mm (16 1/2") catheter with flexible helical coil and 15 openings along the first 40mm / Split needle with puncture needle / Filter 0,2µm / FixoLong |
001158-00Z | 10 |
|
InfiltraLong 500 / 19G x 500mm (19 2/3") catheter with flexible helical coil and 30 openings along the first 75mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
001158-10A | 10 |
|
InfiltraLong 600 / 19G x 600mm (23 2/3") catheter with flexible helical coil and 45 openings along the first 100mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
001158-20D | 10 |
|
InfiltraLong 600 / 19G x 600mm (23 2/3") catheter with flexible helical coil and 60 openings along the first 150mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
001158-20B | 10 |
|
InfiltraLong 700 / 19G x 700mm (27 1/2") catheter with flexible helical coil and 88 openings along the first 220mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
001158-30C | 10 |
|
InfiltraLong 900 / 19G x 900mm (35 2/5") catheter with flexible helical coil and 88 openings along the first 300mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
001158-40C | 10 |
| Item description |
InfiltraLong 420 / 19G x 420mm (16 1/2") catheter with flexible helical coil and 15 openings along the first 40mm / Split needle with puncture needle / Filter 0,2µm / FixoLong |
|
|---|---|---|
| Item no. LUER | 001158-00Z | 10 |
| Item description |
InfiltraLong 500 / 19G x 500mm (19 2/3") catheter with flexible helical coil and 30 openings along the first 75mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
|
| Item no. LUER | 001158-10A | 10 |
| Item description |
InfiltraLong 600 / 19G x 600mm (23 2/3") catheter with flexible helical coil and 45 openings along the first 100mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
|
| Item no. LUER | 001158-20D | 10 |
| Item description |
InfiltraLong 600 / 19G x 600mm (23 2/3") catheter with flexible helical coil and 60 openings along the first 150mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
|
| Item no. LUER | 001158-20B | 10 |
| Item description |
InfiltraLong 700 / 19G x 700mm (27 1/2") catheter with flexible helical coil and 88 openings along the first 220mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
|
| Item no. LUER | 001158-30C | 10 |
| Item description |
InfiltraLong 900 / 19G x 900mm (35 2/5") catheter with flexible helical coil and 88 openings along the first 300mm / Split needle with puncture needle / Filter 0.2µm / FixoLong |
|
| Item no. LUER | 001158-40C | 10 |
| Downloads |
|---|
InFiltra InfiltraLong Brochure
|
Studies:
1 Rawal N., 2012 Perineural catheter analgesia as a routine method after ambulatory surgery – effective but unrealistic, Reg. Anesth. Pain Med. 2012 Jan-Feb; 37(1); 72–76
2 Campolo M., Molin D., Rawal N., Soldati A. Protocols to compare infusion distribution of wound catheters, Med. Eng. Phys. 2011 Apr; 34(3): 326–332
3 Kutzner K. P., Paulini C., Hechtner M., Rehbein P., Pfeil J. Postoperative Analgesie nach Knie-TEP. Intraartikulärer Katheter vs. Nervus-femoralis-Katheter, Der Orthopäde 2015 July; 44(7): 566–573
